Lewis Dot Diagram Of Boron
9.1 Lewis Electron Dot Diagrams
Learning Objective
- Depict a Lewis electron dot diagram for an cantlet or a monatomic ion.
In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. To facilitate our agreement of how valence electrons interact, a elementary way of representing those valence electrons would be useful.
A Lewis electron dot diagramA representation of the valence electrons of an atom that uses dots around the symbol of the element. (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an cantlet that uses dots around the symbol of the chemical element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and beneath the symbol, with no more than two dots on a side. (Information technology does not matter what social club the positions are used.) For example, the Lewis electron dot diagram for hydrogen is simply
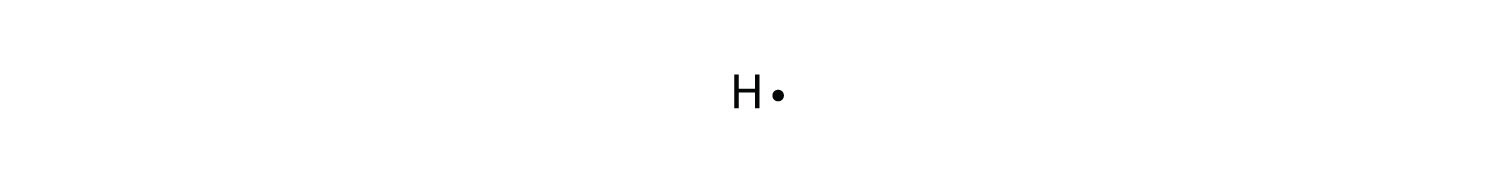
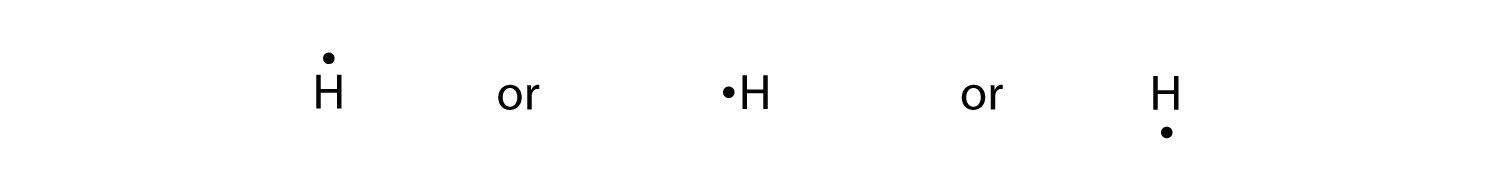
Because the side is not of import, the Lewis electron dot diagram could also be drawn equally follows:
The electron dot diagram for helium, with 2 valence electrons, is as follows:
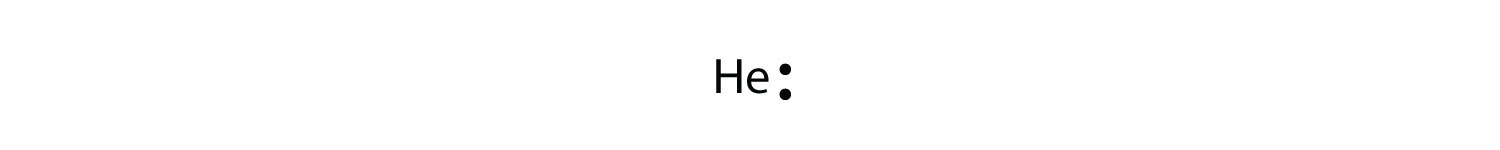
By putting the two electrons together on the same side, we emphasize the fact that these two electrons are both in the ones subshell; this is the common convention we volition adopt, although at that place volition be exceptions subsequently. The adjacent atom, lithium, has an electron configuration of 1s iitwos 1, and so it has only one electron in its valence trounce. Its electron dot diagram resembles that of hydrogen, except the symbol for lithium is used:
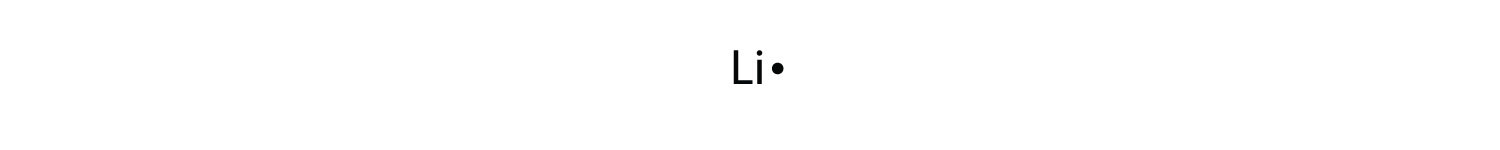
Beryllium has two valence electrons in its 2south shell, and so its electron dot diagram is similar that of helium:

The adjacent atom is boron. Its valence electron shell is 2due south two2p ane, then it has three valence electrons. The third electron will proceed another side of the symbol:
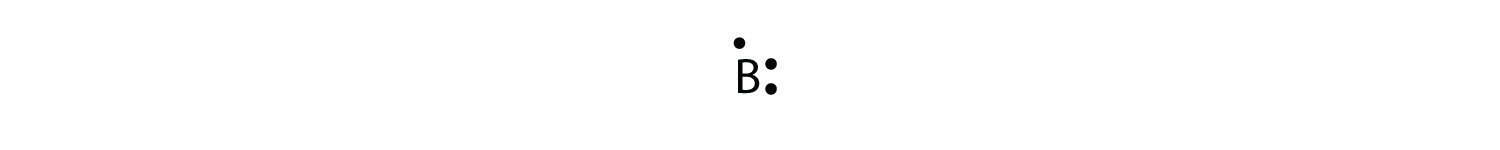
Again, it does not thing on which sides of the symbol the electron dots are positioned.
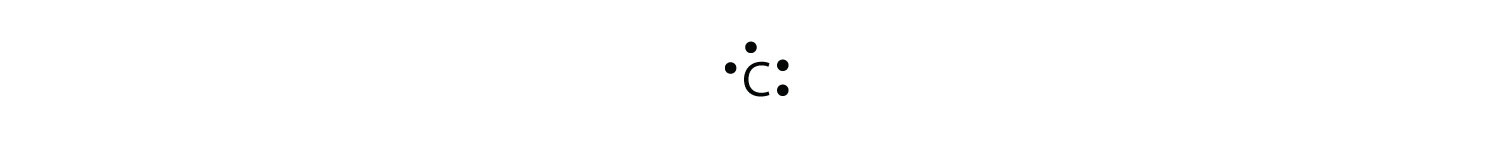
For carbon, there are four valence electrons, two in the 2s subshell and two in the 2p subshell. As usual, we will draw two dots together on one side, to stand for the twos electrons. However, conventionally, we draw the dots for the two p electrons on different sides. As such, the electron dot diagram for carbon is as follows:
With N, which has iii p electrons, we put a single dot on each of the three remaining sides:
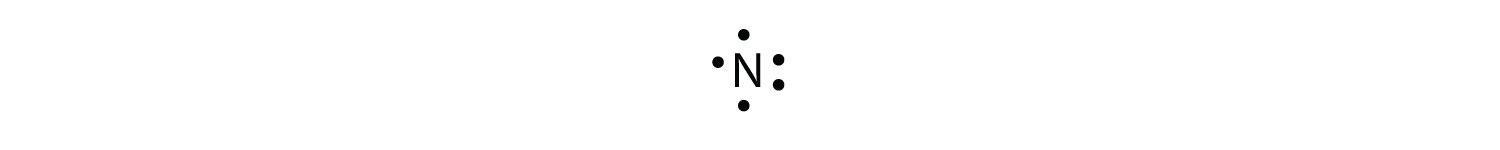
For oxygen, which has 4 p electrons, we now have to start doubling up on the dots on 1 other side of the symbol. When doubling up electrons, make certain that a side has no more than two electrons.

Fluorine and neon have 7 and 8 dots, respectively:

With the side by side element, sodium, the process starts over with a unmarried electron because sodium has a single electron in its highest-numbered shell, the n = 3 shell. By going through the periodic table, nosotros see that the Lewis electron dot diagrams of atoms will never have more than eight dots around the diminutive symbol.
Example 1
What is the Lewis electron dot diagram for each element?
- aluminum
- selenium
Solution
-
The valence electron configuration for aluminum is 3s two3p i. Then it would have iii dots around the symbol for aluminum, two of them paired to represent the 3southward electrons:

-
The valence electron configuration for selenium is fours two4p 4. In the highest-numbered shell, the due north = 4 shell, in that location are six electrons. Its electron dot diagram is as follows:

Examination Yourself
What is the Lewis electron dot diagram for each element?
- phosphorus
- argon
Reply
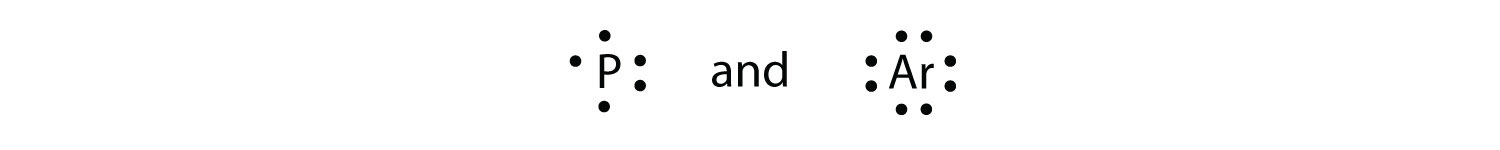
For atoms with partially filled d or f subshells, these electrons are typically omitted from Lewis electron dot diagrams. For example, the electron dot diagram for iron (valence trounce configuration fours 23d vi) is every bit follows:
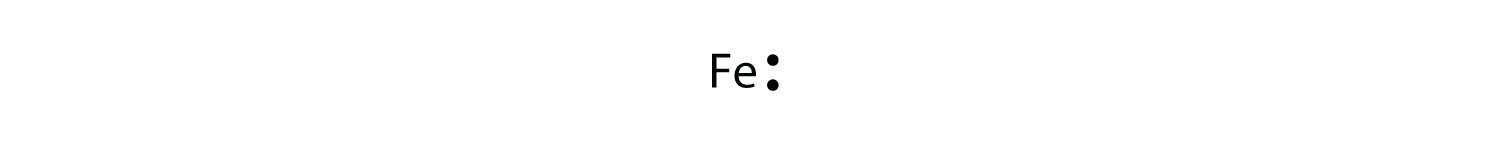
Elements in the same column of the periodic table have like Lewis electron dot diagrams considering they accept the same valence shell electron configuration. Thus the electron dot diagrams for the first column of elements are as follows:

Monatomic ions are atoms that take either lost (for cations) or gained (for anions) electrons. Electron dot diagrams for ions are the same as for atoms, except that some electrons accept been removed for cations, while some electrons take been added for anions. Thus in comparing the electron configurations and electron dot diagrams for the Na atom and the Na+ ion, we annotation that the Na atom has a single valence electron in its Lewis diagram, while the Na+ ion has lost that 1 valence electron:

Technically, the valence vanquish of the Na+ ion is now the north = ii shell, which has viii electrons in it. So why do we not put eight dots around Na+? Conventionally, when nosotros show electron dot diagrams for ions, we show the original valence shell of the atom, which in this case is the n = 3 shell and empty in the Na+ ion.
In making cations, electrons are first lost from the highest numbered shell, not necessarily the last subshell filled. For example, in going from the neutral Fe atom to the Fe2+ ion, the Fe atom loses its two 4s electrons beginning, not its 3d electrons, despite the fact that the 3d subshell is the last subshell beingness filled. Thus we have

Anions have extra electrons when compared to the original atom. Here is a comparing of the Cl cantlet with the Cl− ion:
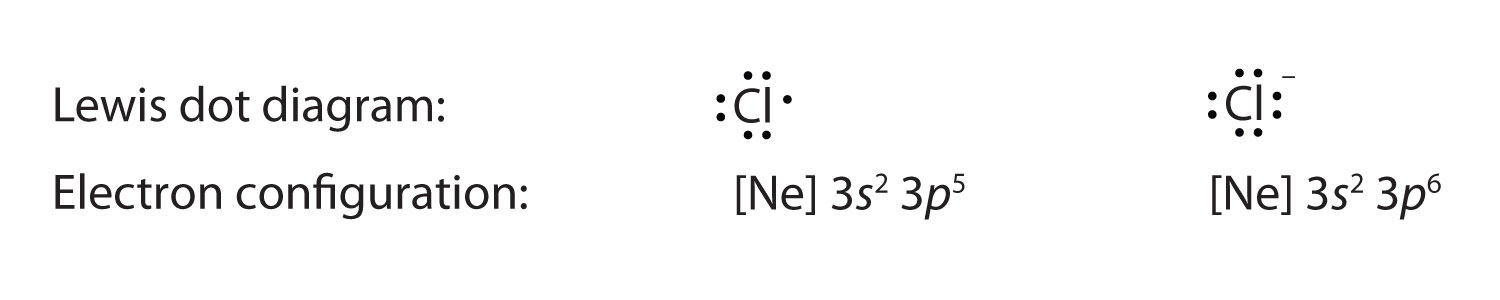
Instance 2
What is the Lewis electron dot diagram for each ion?
- Ca2+
- O2−
Solution
-
Having lost its 2 original valence electrons, the Lewis electron dot diagram is simply Ca2+.
Catwo+ -
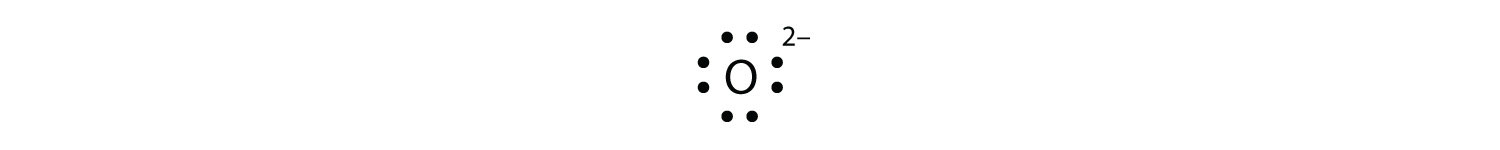
The O2− ion has gained two electrons in its valence shell, so its Lewis electron dot diagram is equally follows:
Examination Yourself
The valence electron configuration of thallium, whose symbol is Tl, is 6southward twovd 106p i. What is the Lewis electron dot diagram for the Tl+ ion?
Reply

Key Takeaways
- Lewis electron dot diagrams utilize dots to represent valence electrons effectually an diminutive symbol.
- Lewis electron dot diagrams for ions take less (for cations) or more than (for anions) dots than the corresponding atom.
Exercises
-
Explain why the showtime two dots in a Lewis electron dot diagram are drawn on the same side of the diminutive symbol.
-
Is it necessary for the showtime dot around an atomic symbol to get on a particular side of the atomic symbol?
-
What column of the periodic table has Lewis electron dot diagrams with two electrons?
-
What cavalcade of the periodic table has Lewis electron dot diagrams that have half-dozen electrons in them?
-
Draw the Lewis electron dot diagram for each element.
- strontium
- silicon
-
Depict the Lewis electron dot diagram for each element.
- krypton
- sulfur
-
Draw the Lewis electron dot diagram for each chemical element.
- titanium
- phosphorus
-
Describe the Lewis electron dot diagram for each element.
- bromine
- gallium
-
Draw the Lewis electron dot diagram for each ion.
- Mg2+
- S2−
-
Depict the Lewis electron dot diagram for each ion.
- In+
- Br−
-
Draw the Lewis electron dot diagram for each ion.
- Atomic number 262+
- N3−
-
Draw the Lewis electron dot diagram for each ion.
- H+
- H−
Answers
-
The showtime two electrons in a valence shell are s electrons, which are paired.
-
the 2nd cavalcade of the periodic table
-
-
-
-
-
- Mg2+
-
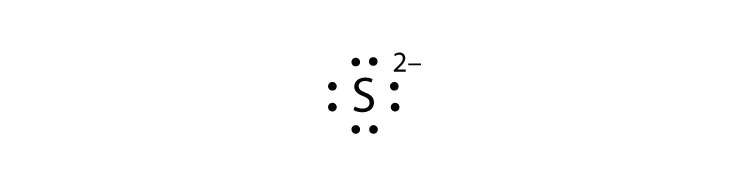
-
- Fe2+
-

Lewis Dot Diagram Of Boron,
Source: https://saylordotorg.github.io/text_introductory-chemistry/s13-01-lewis-electron-dot-diagrams.html
Posted by: middletonbuthrel.blogspot.com


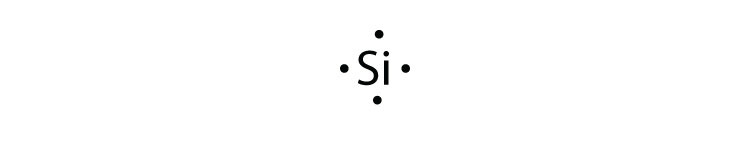

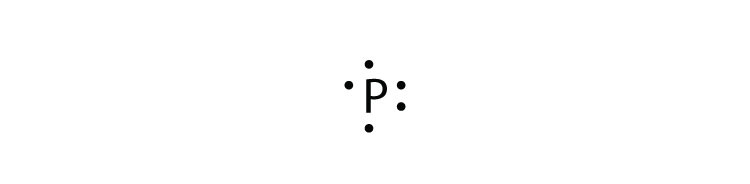

0 Response to "Lewis Dot Diagram Of Boron"
Post a Comment